CDMO Case studies
A trusted CDMO Partner
Galenica has, with our extensive experience and expertise, guided many clients from early pharmaceutical development all the way to approved drug product on the market.
As a reliable contract development partner, we have a proven track record when it comes to quality, knowledge and flexibility as well as keeping timelines.
Development of modified release tablet (Plenadren) for external partner
• DuoCort Pharma, was a Swedish start-up drug development company focused on improving glucocorticoid therapy in areas of significant unmet medical need. The company has undergone a number of changes of ownership over the years and is currently owned by Takeda.
• Duocort found that the current treatment of Addison’s disease with immediate release hydrocortisone products was considered non-optimal both regarding the plasma profile and compliance performance. Galenica was challenged to develop a solid dosage form mimicking the endogenous release profile of cortisol.
Development of modified release tablet (Plenadren) for external partner
• DuoCort Pharma, was a Swedish start-up drug development company focused on improving glucocorticoid therapy in areas of significant unmet medical need. The company has undergone a number of changes of ownership over the years and is currently owned by Takeda.
• Duocort found that the current treatment of Addison’s disease with immediate release hydrocortisone products was considered non-optimal both regarding the plasma profile and compliance performance. Galenica was challenged to develop a solid dosage form mimicking the endogenous release profile of cortisol.
• Galenicas innovative formulation development resulted in a modified release tablet with hydrocortisone in the film coat for initial burst and hydrocortisone in the tablet core for sustained release.
• The phase II/III study in patients confirmed results from the phase I study, demonstrating good clinical effect with one dose per day. The modified release tablet was also well tolerated and safe. The client’s objective was reached and the success factors in this complex pharmaceutical development project was competence, flexibility and transparency, together with the ability to implement the partner’s clinical knowledge into a medical product vital to the patient’s quality of life.
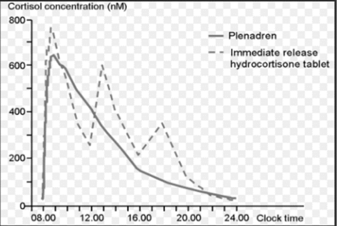
• Galenicas innovative formulation development resulted in a modified release tablet with hydrocortisone in the film coat for initial burst and hydrocortisone in the tablet core for sustained release.
• The phase II/III study in patients confirmed results from the phase I study, demonstrating good clinical effect with one dose per day. The modified release tablet was also well tolerated and safe. The client’s objective was reached and the success factors in this complex pharmaceutical development project was competence, flexibility and transparency, together with the ability to implement the partner’s clinical knowledge into a medical product vital to the patient’s quality of life.
Development of a mometasone furoate cream (Ovixan®) for Galenica
• In 2006 Galenica made a strategic decision to develop its own pharmaceutical products.
• As experts in formulation development, Galenica mapped different drug products where a reformulation could lead to a major improvement in patient compliance. The competitor product Elocon® 0,1 % cream (mometasone furoate) was identified as one suitable candidate.
• Galenica identified and improved the following deficiencies in the original product:
– Reduced the non-compliance criteria of Elocon® cream regarding: Stickiness – Time to rub in – Bad smell
– Improved the cosmetic properties to achieve better patient compliance with maintained efficacy
– Used a modern, eco-friendly, oil-in-water emulsion concept
• A clinical study has confirmed equal efficacy as compared to Elocon cream. An outstanding development of market shares since introduction in 2012 (market share > 70% of mometasone creams in Sweden)
“A Trusted Partner for Inhalation Drug Development”
”Galenica is a highly skilled partner that helps us meet the high-quality standards required for our inhalation drug development projects. It is therefore a natural choice for us to collaborate with Galenica for manufacturing of our clinical trial material, among other things.
Galenica has highly skilled employees that also are excellent to collaborate with throughout the planning and execution stages of projects.”
Johan Wäborg
CEO, Iconovo AB
“Flexible & Competent Partner in GMP Manufacturing”
“Galenica is an important partner for Medicon Valley Inhalation Consortium (MVIC).
MVIC has a broad knowledge base within the development of inhaled medicines and Galenica is a highly competent and flexible partner when it comes to formulation development and GMP manufacturing activities, particularly concerning dry powders for inhalation. During several years, MVIC has performed successful development and manufacturing in inhalation projects together with Galenica.”
Lars Asking
CEO, MVIC AB